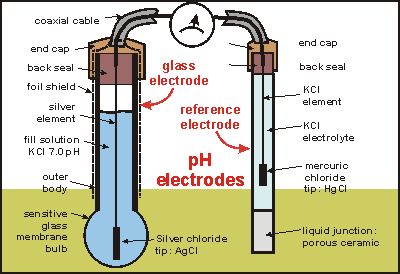
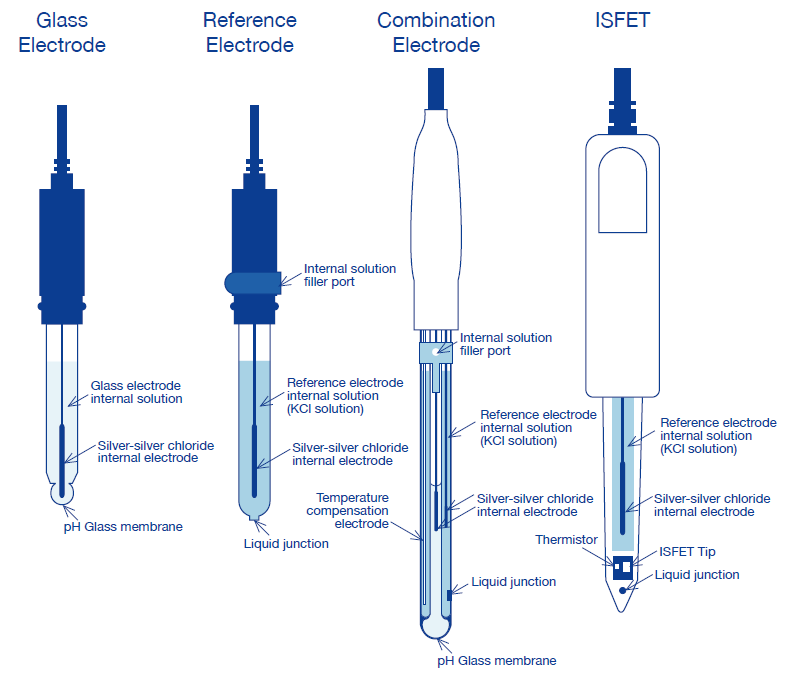
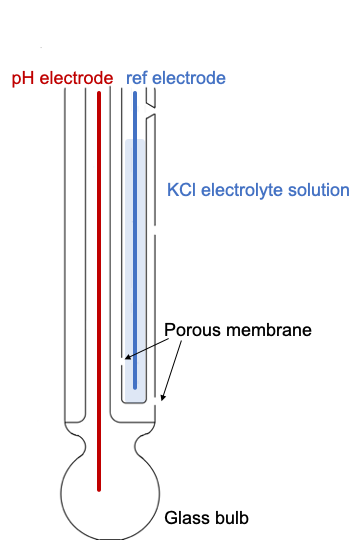
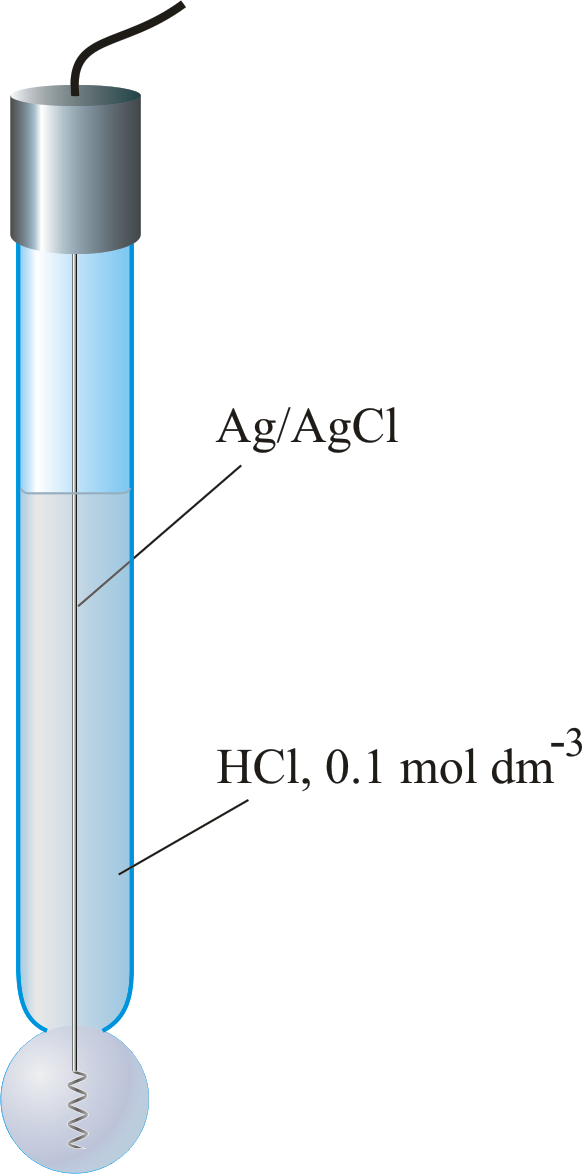
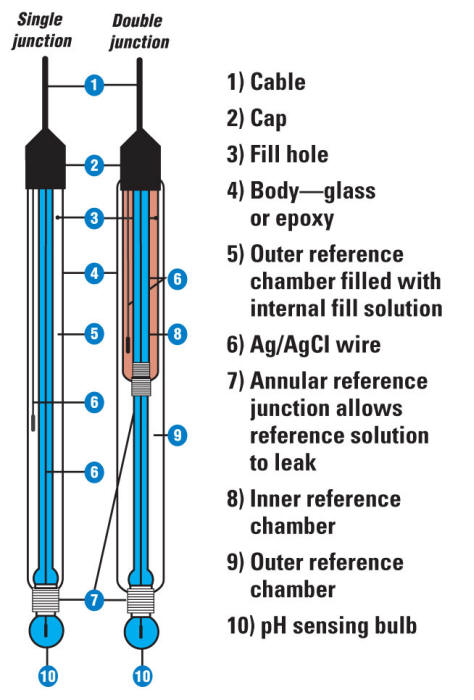
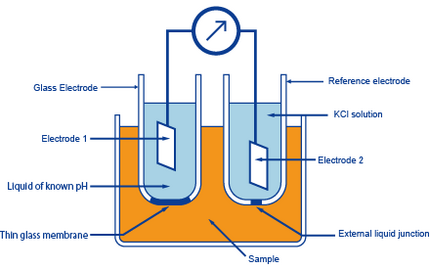
Simple and Economical Procedure To Assemble pH Glass Membrane Electrodes Used in Chemical Education | Journal of Chemical Education

a. Show with the aid of a form of the Nernst equation that when using a pH sensitive glass electrode and reference electrode that the measured potential is proportional to pH. b.