
Suppose a pure Si crystal has 5 × 10^28 atoms m^-3 . it is doped by 1 ppm concentration of pentavalent As. Calculate the number of electrons and holes. Given that n1 = 1.5 × 10^16m^-3 .

Molarity, Molality, Normality, Part per million (ppm) and other basic terms of Concentration solution with definition & formula |Chemistry Basic|02 | by Amrita Shetty | Medium

Phosphotungstic Acid Hydrate, 99.995% (Trace Metals Basis) (Purity excludes up to 300 ppm Si), 10g - Chemsavers, Inc.

▷ La Concentración. max. permisible en ppm de Pb es 0.035 ppm. Si toma una muestra de 850 g de agua ¿Cuál es la cantidad de plomo?

SOLVED: ayuda, esto es de química a) Calcula las ppm de mercurio. Si en 4.3 litros de agua de un rio hay disueltos 0.003g b)Una pasta de dientes tiene 0.004g de floruro
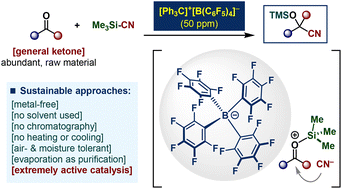
Sustainable organocatalytic cyanosilylation of ketones by PPM-level loading of triphenylcarbenium tetrakis(pentafluorophenyl)borate - Green Chemistry (RSC Publishing)

Solution State Structure Determination of Silicate Oligomers by 29Si NMR Spectroscopy and Molecular Modeling | Journal of the American Chemical Society

















