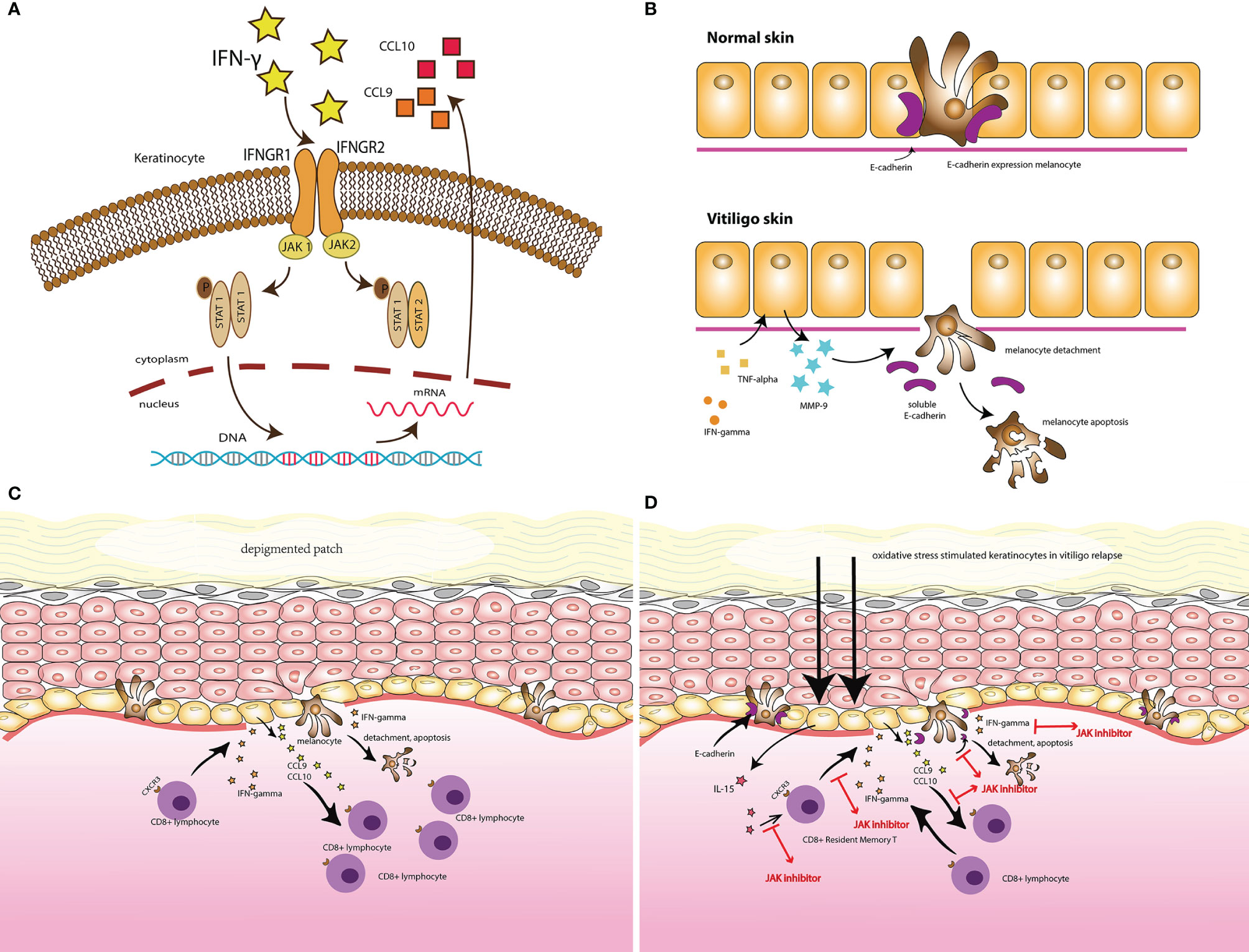
FDA approves JAK inhibitors as 2nd-line systemic therapy in atopic dermatitis < Pharma < Article - KBR

Molecular structures of FDA approved and investigational JAK inhibitors. | Download Scientific Diagram
Pfizer Xeljanz, Tofacitinib (tofacitinib citrate), JAK inhibitor, 11mg tablets, once daily treatment in sustained release tablet form, France Stock Photo - Alamy

Pfizer and Lilly's JAK inhibitor safety concerns prompt Europe to scrutinize drug class | Fierce Pharma

Pfizer Announces FDA Approval of XELJANZ® XR (tofacitinib citrate) Extended-Release Tablets, the First and Only Once-Daily Oral JAK Inhibitor Treatment for Rheumatoid Arthritis | Business Wire
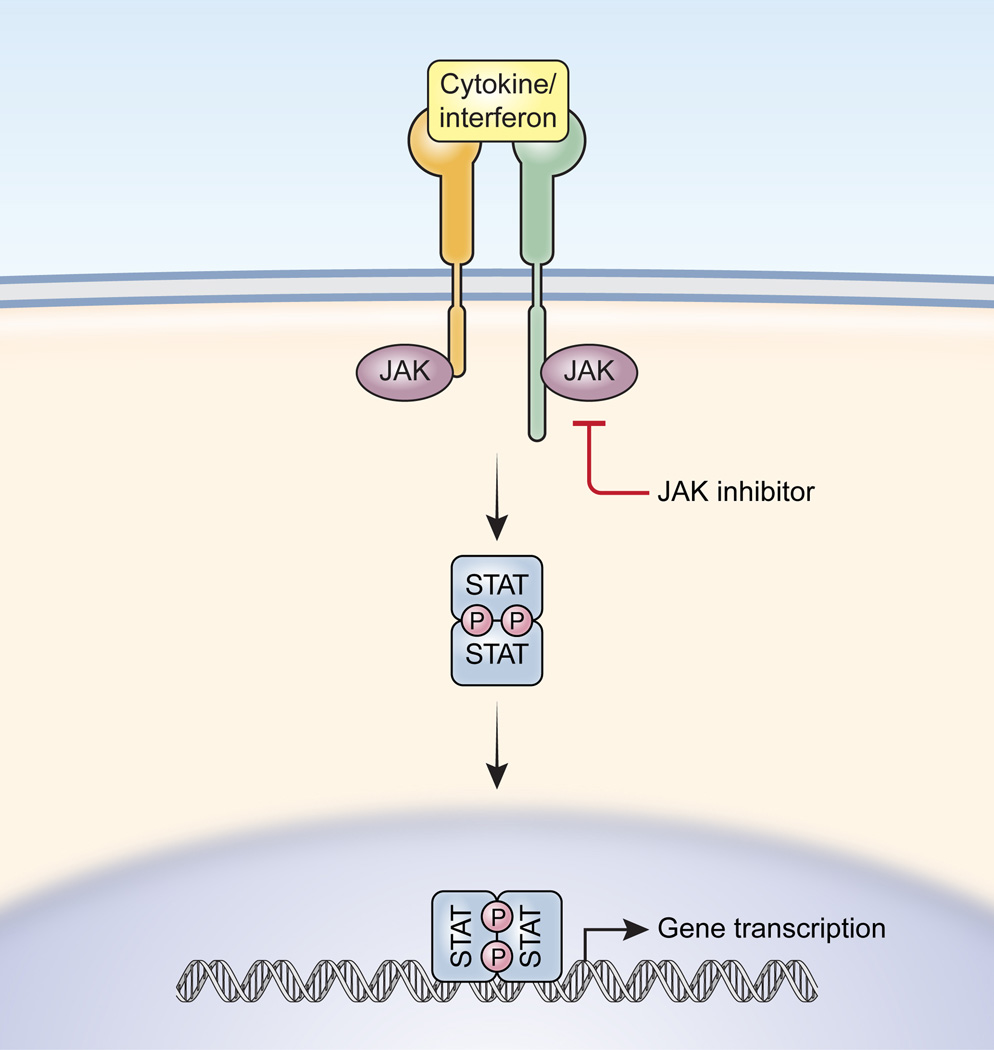
FDA Crisis for Pfizer: The Impact of an FDA Warning On the Company – Cases and Tools in Biotechnology Management

Janus kinase-targeting therapies in rheumatology: a mechanisms-based approach | Nature Reviews Rheumatology
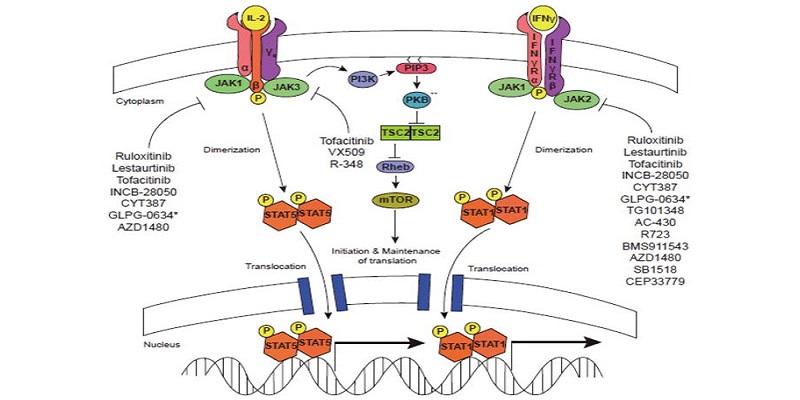
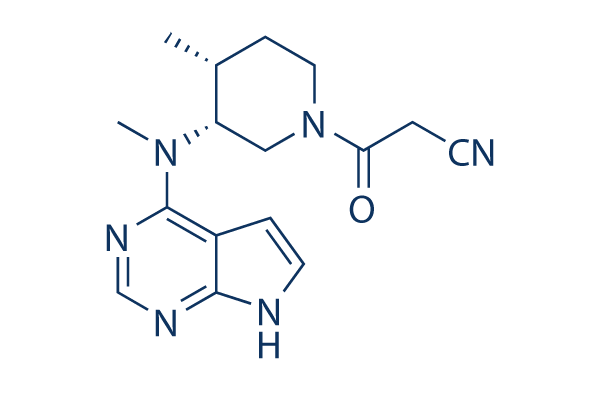


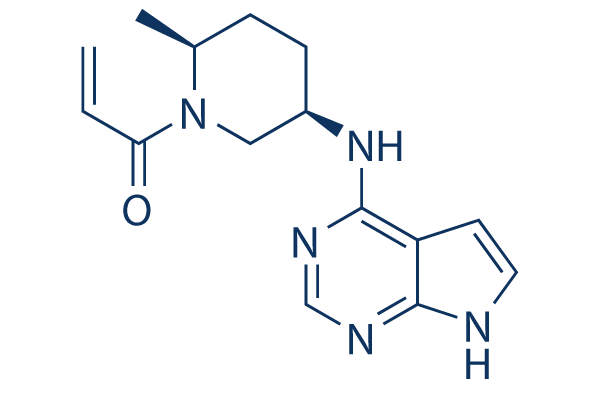

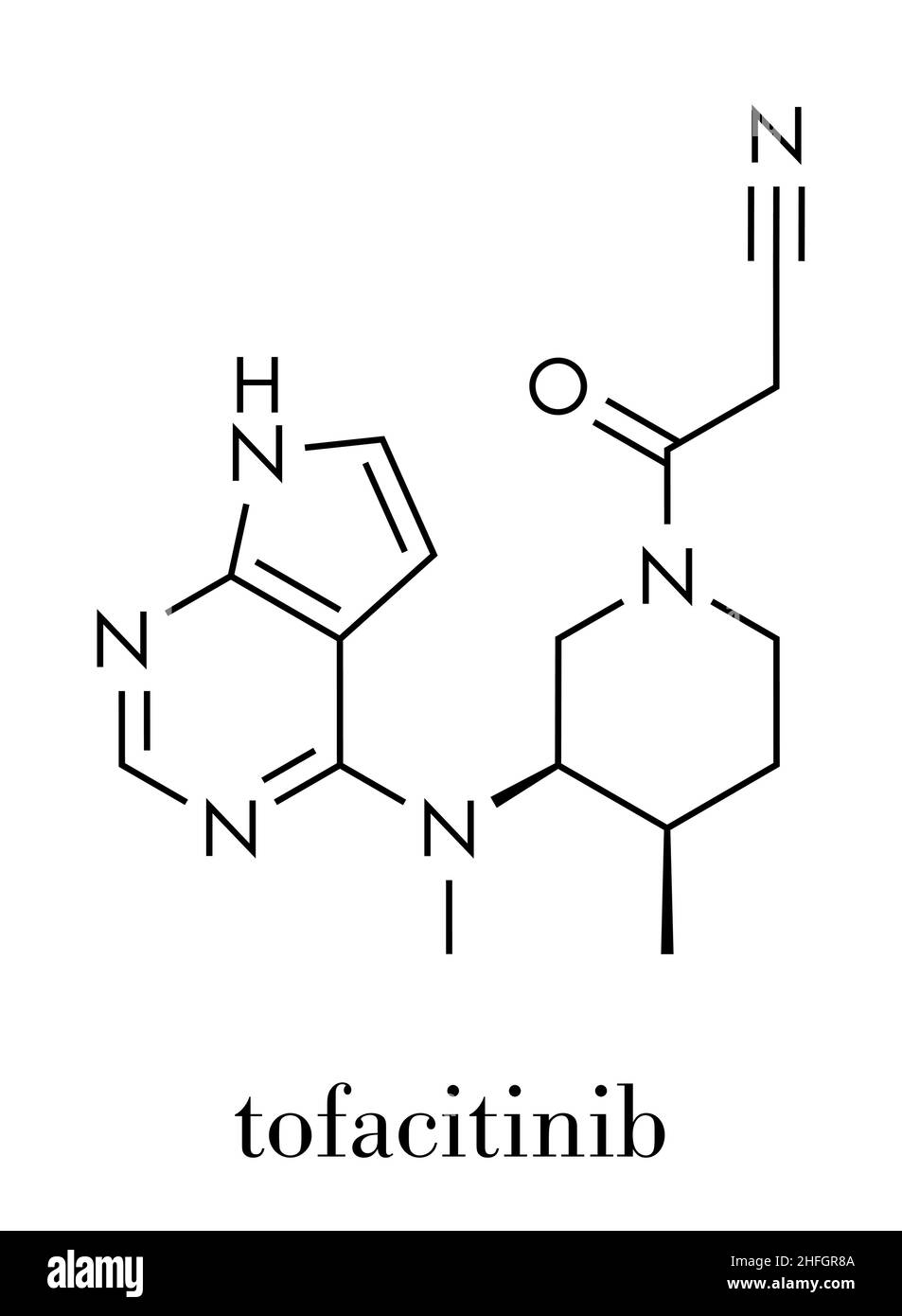


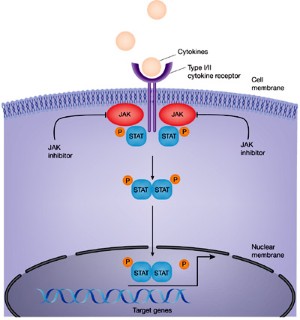


:max_bytes(150000):strip_icc()/jak-inhibitors-4706526-FINAL-3461d4fc6ce44f54801ef59489afa62c.png)