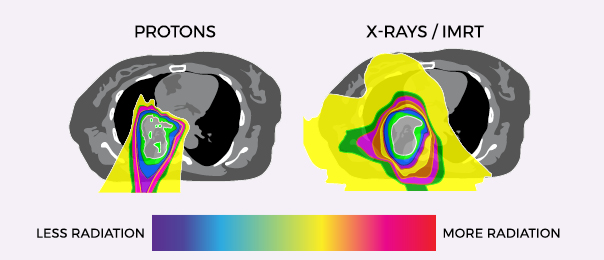
Lung Cancer Treatment Program | Thoracic Oncology Program | Thoracic Cancer Program | Rutgers Cancer Institute of NJ

Lung Cancer Survivors On Long Island Rave About Immunotherapy, Which Doctors Say Is Akin To A Cure For Some - CBS New York

Lung Cancer Treatment Program | Thoracic Oncology Program | Thoracic Cancer Program | Rutgers Cancer Institute of NJ

Cemiplimab monotherapy for first-line treatment of advanced non-small-cell lung cancer with PD-L1 of at least 50%: a multicentre, open-label, global, phase 3, randomised, controlled trial - The Lancet

First in New Jersey, and One of First in Nation, to Use New Technology for Treatment of Lung Cancer - Englewood Health

Cooperman Barnabas Medical Center - As Lung Cancer Awareness Month comes to an end, we want to remind you that #LungCancer is the leading cause of cancer death in the United States.