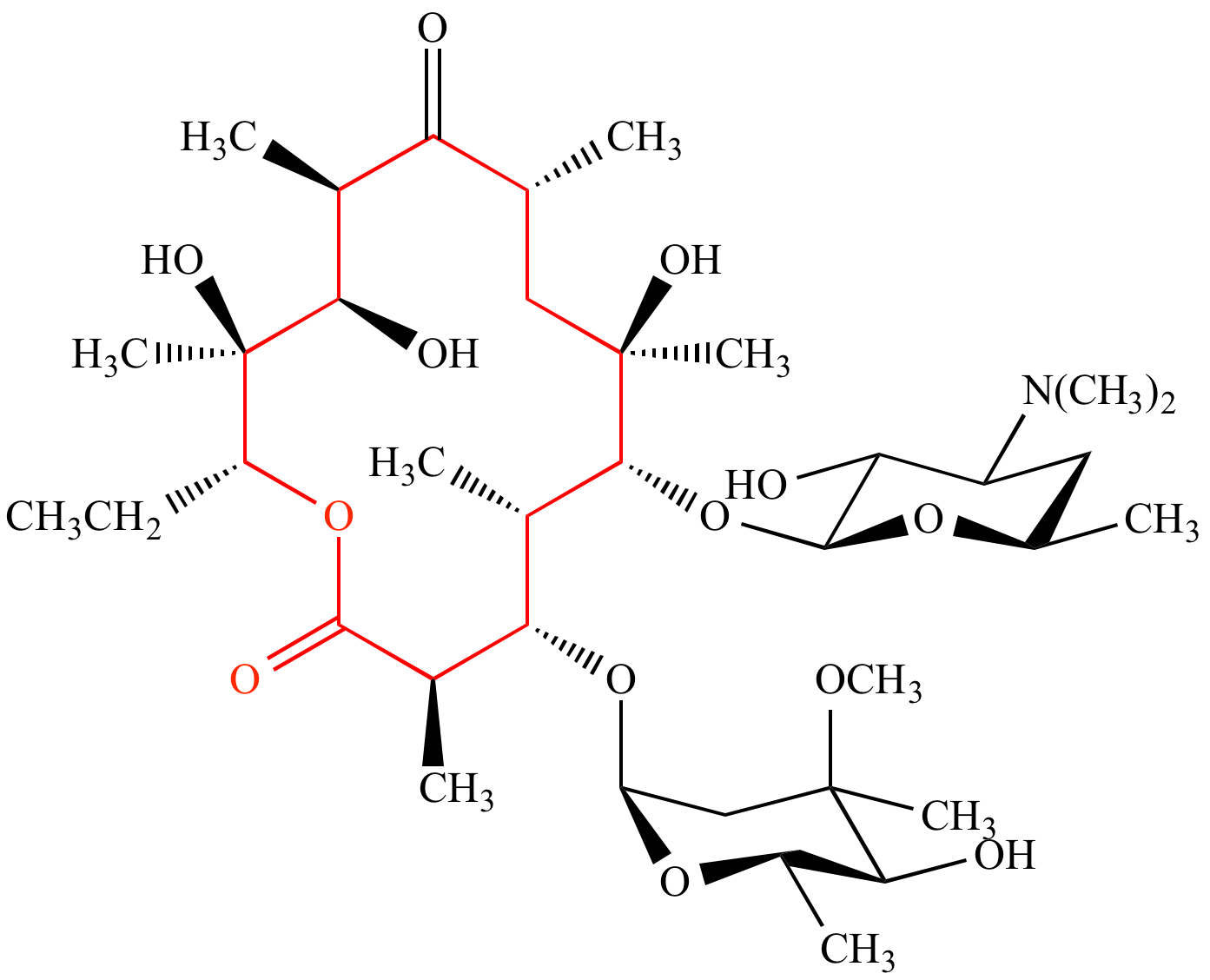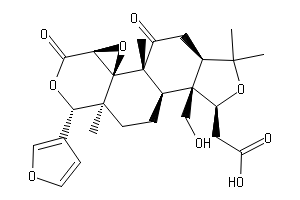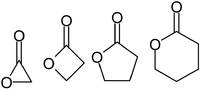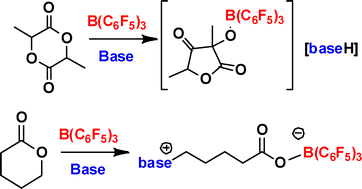
Ring openings of lactone and ring contractions of lactide by frustrated Lewis pairs - Dalton Transactions (RSC Publishing)

Size range of lactone rings produced by TE domains. The lactone ring of... | Download Scientific Diagram

the Sugar and Lactone Ring Structure-Activity Relationships for the Hypertensinogenic Activity of Ouabain : Role of | Semantic Scholar

Mild and Chemoselective Lactone Ring-Opening with (TMS)ONa. Mechanistic Studies and Application to Sweroside Derivatives | The Journal of Organic Chemistry

Organocatalysis applied to the ring‐opening polymerization of β‐lactones: A brief overview - Khalil - 2019 - Journal of Polymer Science Part A: Polymer Chemistry - Wiley Online Library

A thermal lactone ring opening/closing reaction. H-EA=Electron acceptor | Download Scientific Diagram

Active parts of aflatoxin molecule Key; a -Furan ring, b-lactone ring,... | Download Scientific Diagram

Synthesis of New Camptothecin Analogues with the E‐Lactone Ring Replaced by α,β‐Cyclohexenone - Bacherikov - 2006 - European Journal of Organic Chemistry - Wiley Online Library

Catalytic Redox Chain Ring Opening of Lactones with Quinones To Synthesize Quinone-Containing Carboxylic Acids | Organic Letters
Ring-opening polymerization of a five-membered lactone trans-fused to a cyclohexane ring | Polymer Journal
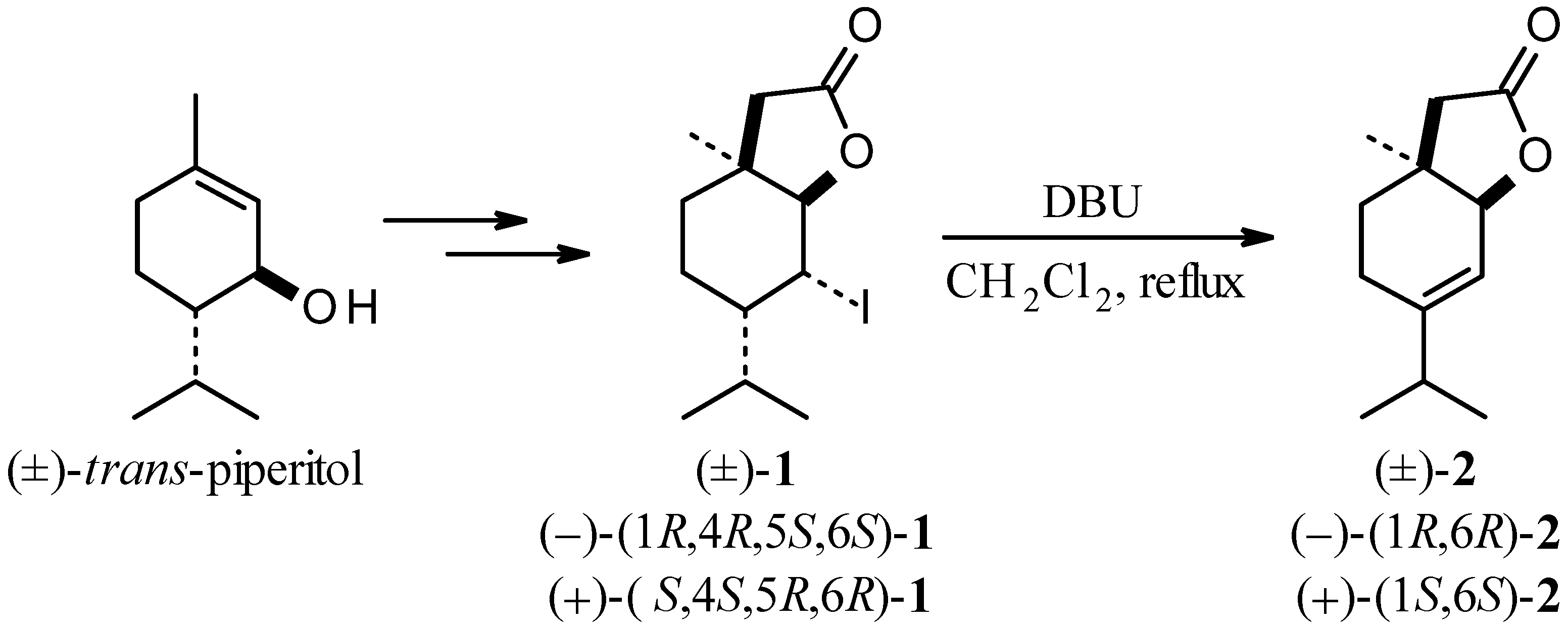
Molecules | Free Full-Text | Lactones 41. Synthesis and Microbial Hydroxylation of Unsaturated Terpenoid Lactones with p-Menthane Ring Systems