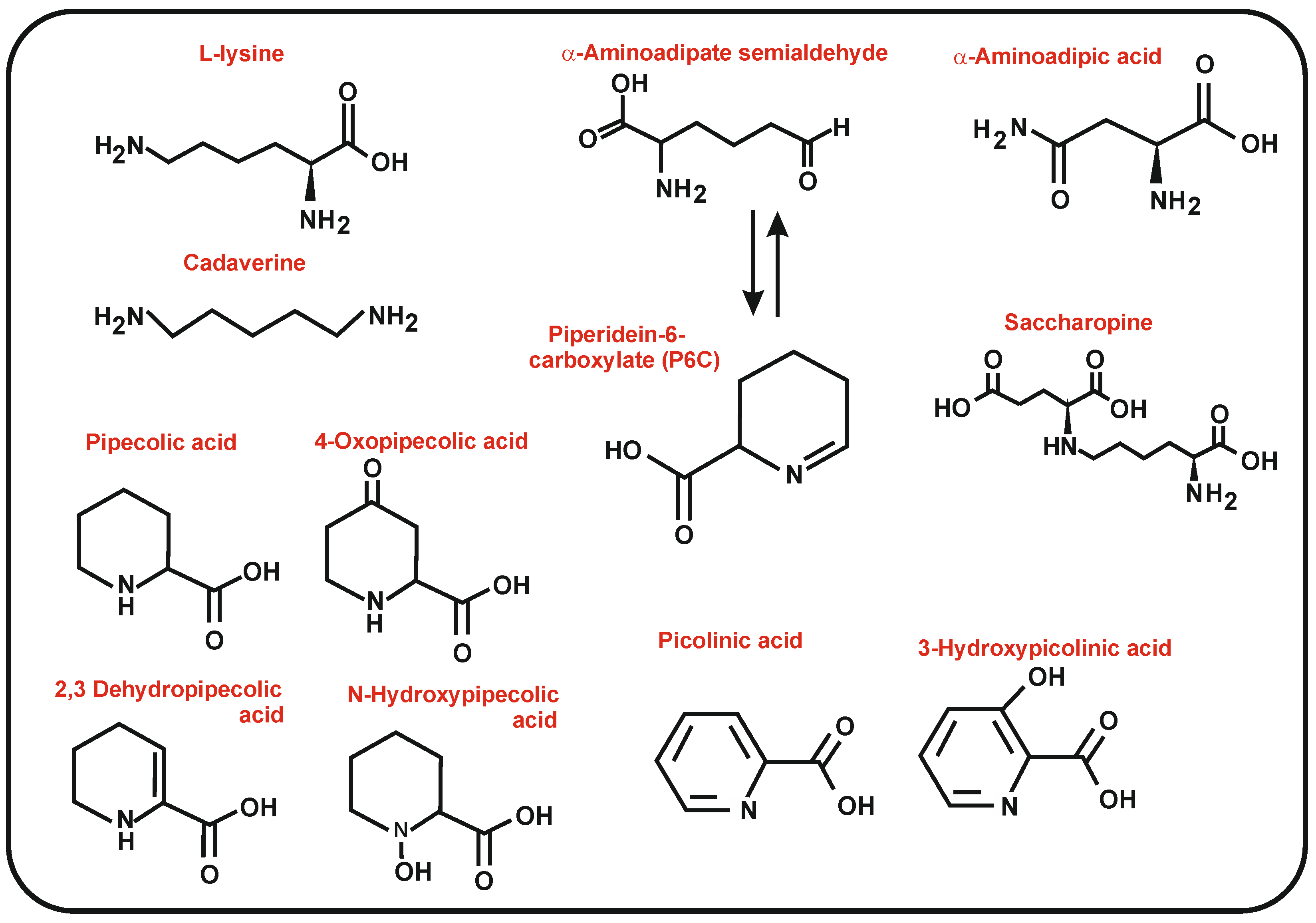
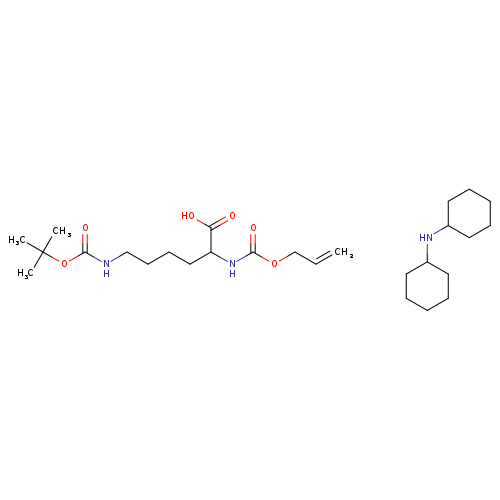
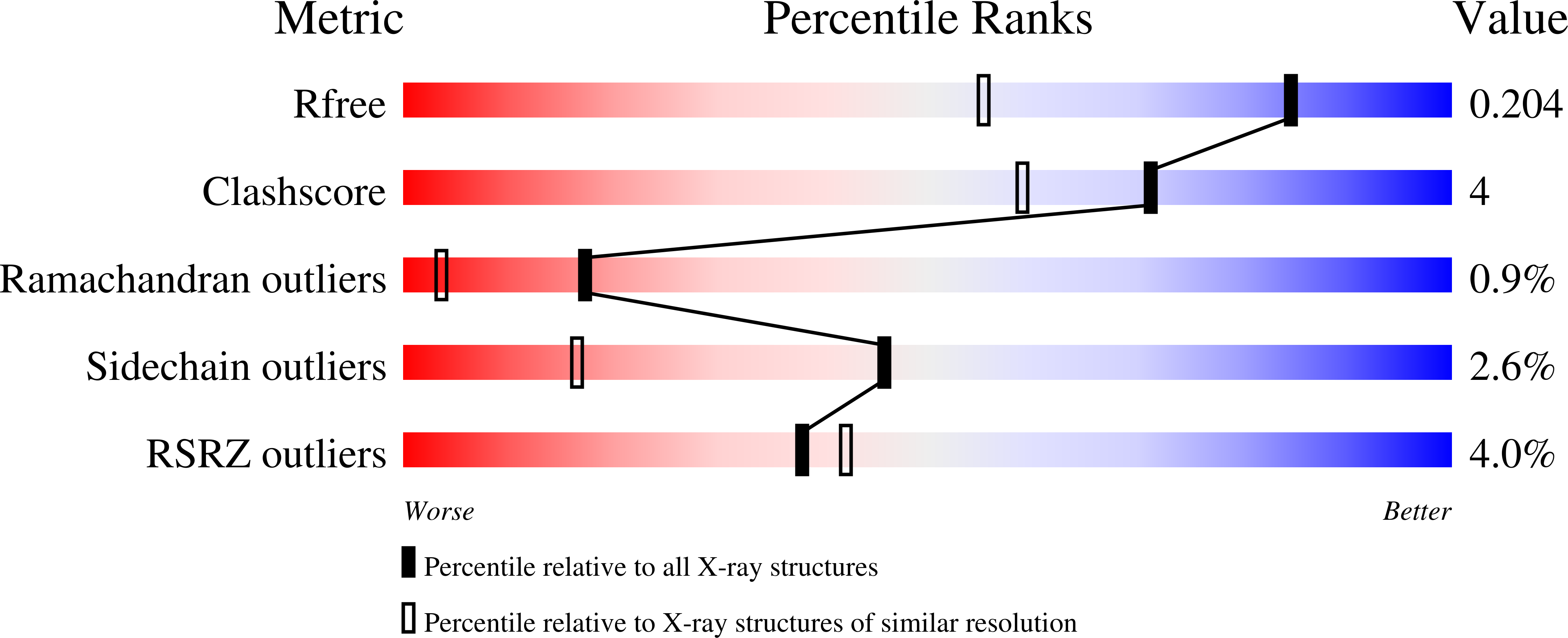
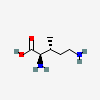Molecules | Free Full-Text | GC-MS Discrimination of Citrulline from Ornithine and Homocitrulline from Lysine by Chemical Derivatization: Evidence of Formation of N5-Carboxy-ornithine and N6-Carboxy-lysine
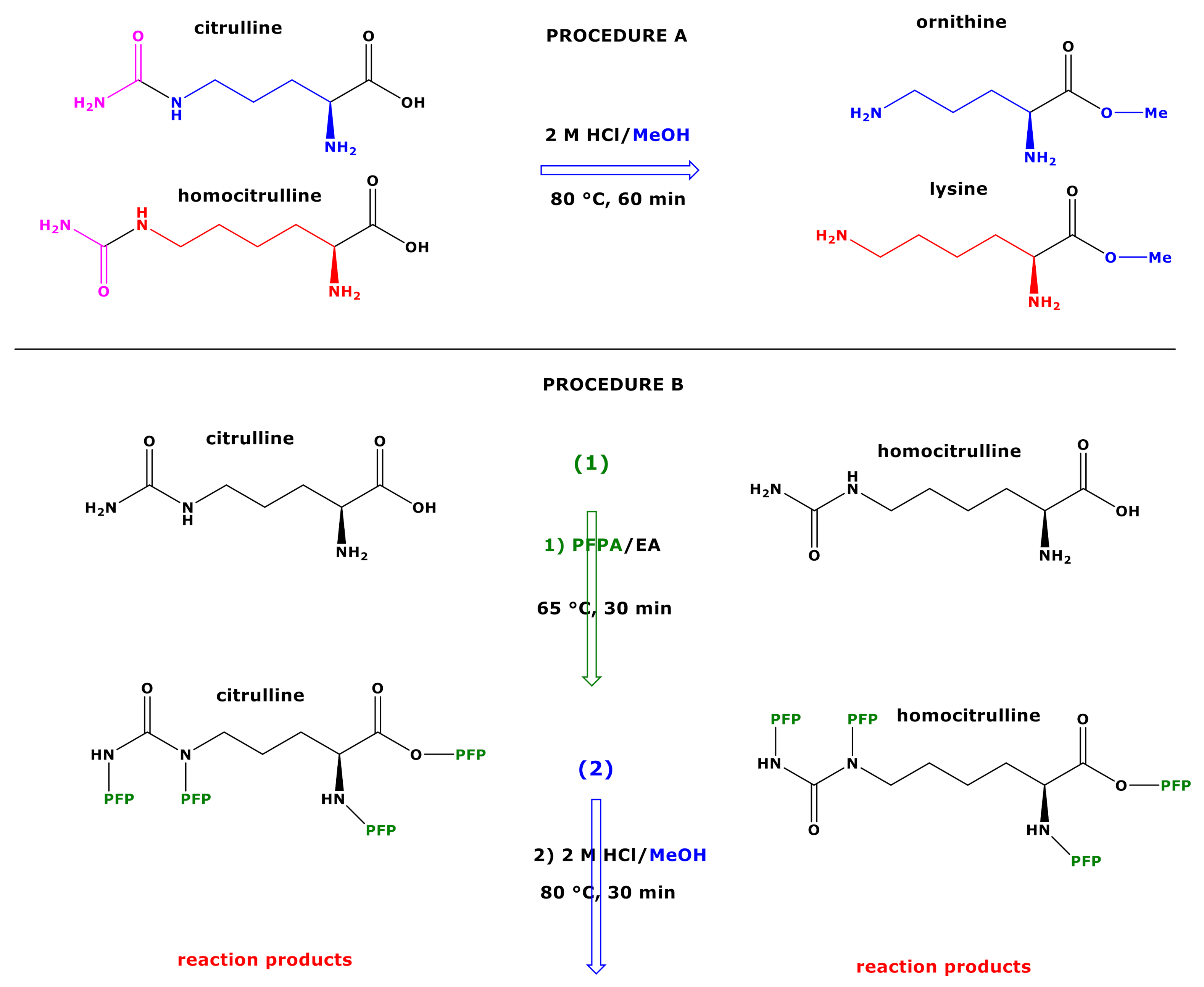
Molecules | Free Full-Text | GC-MS Discrimination of Citrulline from Ornithine and Homocitrulline from Lysine by Chemical Derivatization: Evidence of Formation of N5-Carboxy-ornithine and N6-Carboxy-lysine

Therapeutic Potential of Foldamers: From Chemical Biology Tools To Drug Candidates? | Journal of Medicinal Chemistry

Structure and Reaction Mechanism of Pyrrolysine Synthase (PylD) - Quitterer - 2013 - Angewandte Chemie International Edition - Wiley Online Library

Biosynthesis of the 22nd Genetically Encoded Amino Acid Pyrrolysine: Structure and Reaction Mechanism of PylC at 1.5 Å Resolution - ScienceDirect

Molecules | Free Full-Text | Structural and Mechanistic Basis for the Inactivation of Human Ornithine Aminotransferase by (3S,4S)-3-Amino-4-fluorocyclopentenecarboxylic Acid

Evaluation of Metalloendopeptidase Lys-N Protease Performance under Different Sample Handling Conditions | Journal of Proteome Research